When we walk into any modern home, laboratory, or industrial setup, we will find one technology quietly transforming electrical energy into visible brilliance—the Light Emitting Diode (LED). From smartphone screens and traffic signals to advanced optical communication systems, LEDs have become an indispensable part of engineering life.
At its core, a light emitting diode is a two-terminal semiconductor optoelectronic device that converts electrical energy directly into light through a fascinating quantum mechanical process called electroluminescence. Unlike conventional bulbs that waste energy as heat, LEDs are efficient, compact, and long-lasting.
In this article, you will learn:
- The physics behind light emission from semiconductors
- How LEDs work at the atomic level
- Mathematical understanding of photon energy
- Practical applications and engineering relevance
Let us begin by tracing how this remarkable device came into existence.
Contents
Toggle🔶 Historical Background of LED
The story of the LED begins in the early 20th century. In 1907, H.J. Round, a British experimenter, observed electroluminescence in silicon carbide. However, it remained a scientific curiosity for decades.
The first practical LED was developed in 1962 by Nick Holonyak Jr., often called the “father of the LED,” who created a visible red LED using gallium arsenide phosphide (GaAsP).
Since then, LED technology has evolved dramatically:
- 1970s: Low-intensity red and green LEDs
- 1990s: Blue LED breakthrough (enabled white LEDs)
- Today: High-efficiency LEDs are used in lighting, displays, and communication
This evolution reflects how fundamental semiconductor physics can transform industries.
🔶 Main Concept: What is a Light Emitting Diode?
A Light Emitting Diode (LED) is a semiconductor device that emits light when it is forward-biased.
Basic Principle of LED
The operation of an LED is based on the principle of electroluminescence.
Electroluminescence is the phenomenon in which a material emits light when an electric current or electric field passes through it, due to the recombination of charge carriers.
What Happens Physically in an LED?
When a forward voltage is applied:
- Electrons from the n-side move toward the p-side.
- Holes from the p-side move toward the n-side.
- At the junction, electrons recombine with holes.
- During recombination, the excess energy is released in the form of photons, which appear as visible light.
👉 Think of it like this:
Electrons are like excited travelers moving from a higher energy level to a lower one. When they “settle down,” they release energy—not as heat, but as light.
Symbol of LED
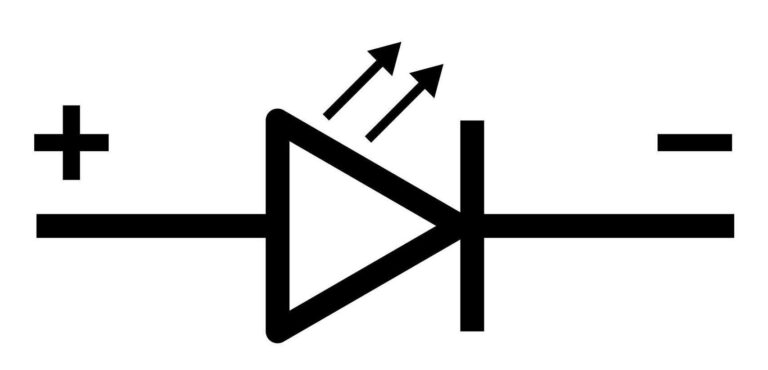
🔶 Construction & Structure of LED
Light Emitting Diodes are fabricated using direct bandgap semiconductor materials such as Gallium Arsenide (GaAs), Gallium Phosphide (GaP), and Gallium Nitride (GaN), because these materials can efficiently convert electrical energy into visible light during electron–hole recombination.
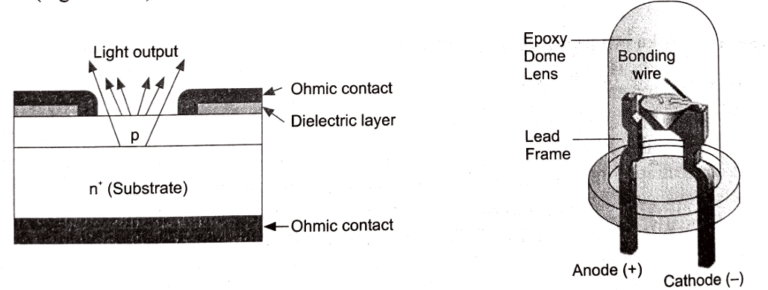
Structurally, an LED consists of a p-type layer and an n-type layer forming a junction, with a depletion region at their interface where recombination occurs, all enclosed within a transparent casing that allows the emitted light to pass out effectively.
The construction of a surface-emitting LED is shown in the following figure. An n-type layer is grown on a substrate, and a p-type layer is grown on it by the process of diffusion. The p-layer is made very thin to prevent loss of photons due to absorption in the layer. Metal connections are made at the edges of the p-layer to allow more central surface for the light to escape.
A metal film is deposited at the bottom of the substrate for reflecting as much light as possible towards the surface of the device and also to provide an electrode connection.
The light generated at the junction may not emerge from the surface of the device, as it is likely to suffer total internal reflection at the semiconductor-air boundary. Therefore, the device is encapsulated in a clear epoxy resin of suitable refractive index.
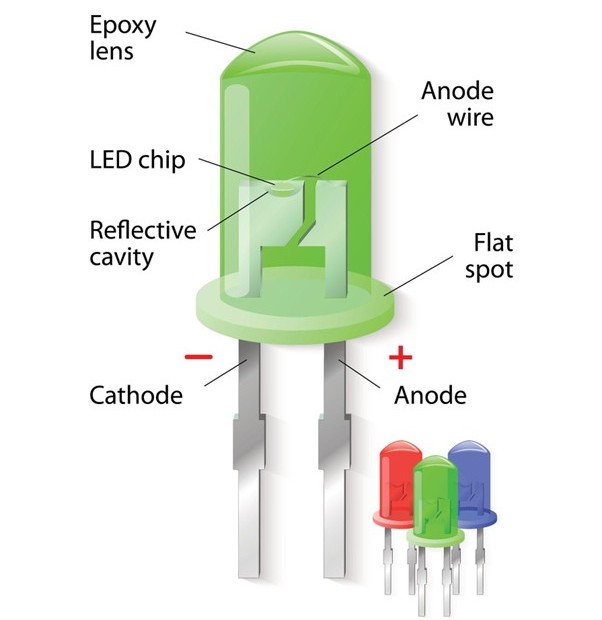
Active region:
In practical LEDs, light is mainly produced in a small region near the junction called the active region, where the concentration of electrons and holes is highest, and recombination occurs most efficiently.
In modern high-brightness LEDs, this region is often designed as a quantum well, which is an extremely thin layer of lower bandgap material placed between higher bandgap layers.
This structure confines charge carriers in a small space, increasing their chances of recombination and thereby enhancing light output efficiency.
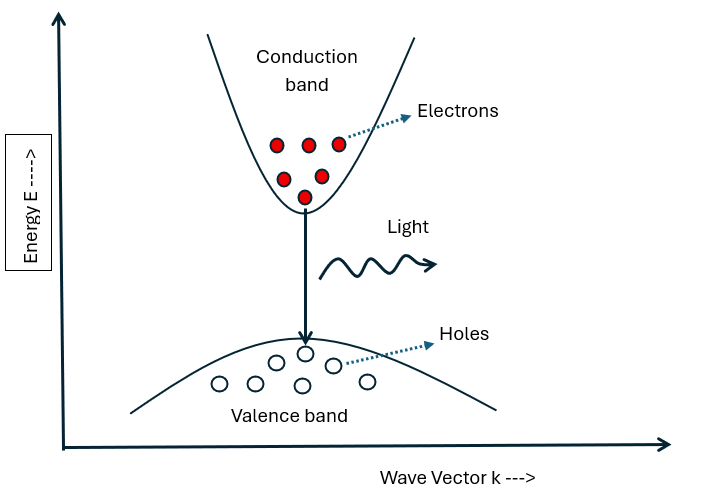
🔶 Energy Band Diagram of Light Emitting Diode
An energy band diagram represents the distribution of energy levels available to electrons in a solid, mainly showing the valence band, the conduction band, and the band gap between them.
It helps in understanding how electrons move and participate in conduction or light emission. In devices like LEDs, it explains how electrons transition from higher to lower energy levels and release energy in the form of photons.
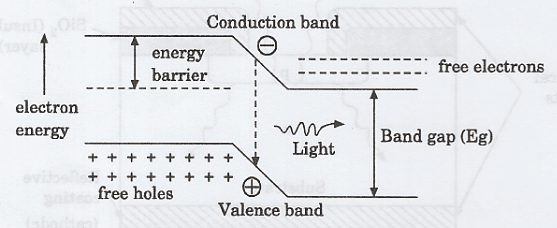
🔶 Working of Light Emitting Diode
When a Light Emitting Diode (LED) is forward biased, the applied voltage reduces the potential barrier at the junction, allowing electrons from the n-region and holes from the p-region to move across into opposite sides.
These charge carriers, now acting as minority carriers, diffuse away from the junction and eventually encounter carriers of opposite charge. When an electron recombines with a hole in a direct bandgap semiconductor, it loses energy by dropping from a higher energy level to a lower one, and this energy is released in the form of a photon.
The energy of the emitted photon is approximately equal to the bandgap energy, so the wavelength—and hence the color—of the light depends on the material used in the LED.
🔶 Colour of Emitted Light
The wavelength of emitted light depends on the band gap: $$\lambda=\frac{hc}{E_g}$$ Hence,
- Larger band gap → shorter wavelength (blue light)
- Smaller band gap → longer wavelength (red light)
🔶 Applications of Light Emitting Diode
- Display Technology: LEDs are widely used in televisions, smartphones, and large display boards because they provide high brightness, excellent efficiency, and long-lasting performance.
- Optical Communication: LEDs are employed in fiber optic systems to transmit information in the form of light signals over long distances.
- Traffic Signals: LEDs are ideal for traffic lights due to their long lifespan and low power consumption, making them both reliable and energy-efficient.
- Medical Devices: LEDs are used in medical applications such as phototherapy treatments and diagnostic equipment because of their precise and controlled light emission.
- Automotive Lighting: Modern vehicles use LEDs extensively in headlights, tail lights, and dashboard indicators due to their durability, brightness, and energy efficiency.
🔶 Conclusion
Let’s review the main conclusions:
- A Light Emitting Diode converts electrical energy into light.
- It works on electron-hole recombination.
- LEDs are long-lasting, small, and effective.
- They are essential to contemporary communication and electronics.
Hence, LEDs are a stunning example of how modern engineering is directly powered by quantum physics. They are the ideal fusion of theory and practice, ranging from small indicators to enormous displays.
🔶 Important Questions for Exam Preparation
- What is a light-emitting diode? Explain its working principle with the help of an energy band diagram.
- Distinguish between a direct bandgap semiconductor and an indirect bandgap semiconductor. Why can silicon not be used as an efficient LED material?
- Why are direct bandgap semiconductors used in LEDs?
- Compare an LED with an ordinary p–n junction diode.
🔶 FAQs
Q. Which materials are used in LEDs?
Gallium arsenide (GaAs), gallium nitride (GaN), and related compounds.
Q. What is a Light Emitting Diode?
A Light Emitting Diode is a semiconductor device that emits light when current flows through it.
Q. Why do LEDs emit light?
Because electrons recombine with holes and release energy as photons.
Q. Why are LEDs energy efficient?
They convert most electrical energy into light instead of heat.
Q. What determines LED color?
The band gap energy of the semiconductor material.

I recently started looking into upgrading my laptop’s memory. My
research eventually led me to https://znayka.com.ua.
I couldn’t find practical upgrade instructions on most websites. Many sources were missing key details.
On this site I found clear explanations about RAM compatibility, along with step-by-step visuals that made everything much easier to understand.
I recommend this site to anyone looking for clear tech explanations if you want reliable information about laptops and hardware upgrades.
A car accident happened and I needed to act quickly.
I started searching for legal information online.
https://legaltelegraph.com
It was hard to find honest legal answers.
I found the guidance I needed.
It was a huge support.